Featured news
All news

London Marathon runners raise a record-breaking £85,000 for leading hospital charity
Runners in this year’s TCS London Marathon have broken an all-time record for Addenbrooke’s Charitable Trust (ACT) – raising £85,000 for the new Cambridge Cancer Research Hospital and Cambridge Children's Hospital – with just 19 runners!

High Sheriff showcases contribution of staff and patients shaping the future Cambridge Cancer Research Hospital
The recently appointed High Sheriff of Cambridgeshire has visited the site of the new Cambridge Cancer Research Hospital yesterday (20 April) to highlight plans and discover how the project is progressing.

Cambs and Peterborough patients to benefit from new self-referral breast cancer service
Patients from Cambridgeshire and Peterborough with worrying breast cancer symptoms can now be referred directly to specialist clinics in hospital, without needing a GP appointment.

Cambridge Dragon Boat Festival launches raising funds for new Cancer Hospital
The Cambridge Dragon Boat 2026 Festival has officially launched to celebrate its 20th year – with Addenbrooke's Charitable Trust promising everyone who takes part will make a ‘meaningful difference’ to cancer care in the region, by raising funds for Cambr

ACT announce 105K raised at 2026 Cambridge Half Marathon for Cambridge Cancer Research Hospital
The Cambridge Cancer Research Hospital team and Addenbrooke's Charitable Trust have extended their heartfelt thanks to everyone who took part in this year’s Cambridge Half Marathon, celebrating the incredible efforts of runners who helped raise over £105,000 for the new Cancer Hospital.

Join us at Cambridge Festival and get hands-on with cancer research in Cambridge
Join us at Cambridge Festival on Saturday 21 March 2026 from 11am – 4pm at the Cambridge Academy for Science and Technology (CAST) on the Cambridge Biomedical Campus, to learn more about our plans to build Cambridge Cancer Research Hospital.

Professor Rebecca Fitzgerald elected as Fellow of AACR
The Research Lead for Cambridge Cancer Research Hospital, Professor Rebecca Fitzgerald, is elected as a 2026 Fellow of the American Association for Cancer Research.

Hot flush treatment has anti-breast cancer activity, study finds
A drug mimicking the hormone progesterone has anti-cancer activity when used together with conventional anti-oestrogen treatment for women with breast cancer, a new Cambridge-led trial has found.

Behind the scenes: Stand Up To Cancer live from Addenbrooke’s
Last week a special programme for Stand Up To Cancer presented by Davina McCall was broadcast live from Addenbrooke’s Hospital.

Collaboration with new Digital Health and Surgical Training Centre
Carrie Symington, Senior Improvement Manager, and Elaine Chapman, Lead Nurse for the Cambridge Cancer Research Hospital (CCRH) project recently had the pleasure of visiting the Cambridge Surgical Training Centre (CSTC) on the Quorum site. They discuss what the new area will offer staff across the region, what they learned from the tour, and the opportunities the Centre presents for staff working in the future Cancer hospital.

Pioneering trial offers hope on world pancreatic cancer day
A pioneering trial could improve treatment for late-stage pancreatic cancer by reducing side effects from chemotherapy.

Addenbrooke’s to host live Channel 4 programme for Stand Up To Cancer
A pioneering Channel 4 programme presented by Davina McCall will be broadcast live from Addenbrooke’s Hospital as part of this year’s Stand Up To Cancer 2025. It will highlight the expertise, innovation and dedication of clinicians in Cambridge, as 'Cancer Clinic: Live' broadcasts from a special one-off cancer clinic at Addenbrooke’s on the evening of Friday 12th December.
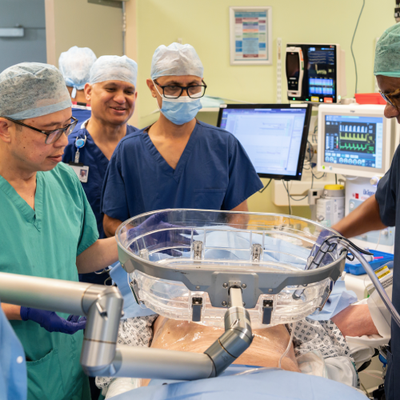
Addenbrooke’s begins innovative liver cancer treatment for first NHS patients in Europe
Cambridge University Hospitals NHS Foundation Trust (CUH) has become the first hospital in Europe to deliver histotripsy treatment to a patient outside of a clinical trial, after being fast-tracked by the government - marking a major milestone in NHS cancer care.

Pioneering one-stop kidney clinic provides same day diagnosis and halves cancer waiting times
In a UK first, a one-stop kidney cancer clinic at Addenbrooke’s Hospital is pioneering an ultra-fast way of diagnosing cancer, which is cutting waiting times for patients by a month.

Trust clinicians to feature in Cambridge Film Festival
Shades of Survival will be shown on 26 & 28 October at the Cambridge Film Festival, exploring inequalities in breast cancer care for Black women.

Cambridge Dragon Boat Festival raises vital funds for Cambridge Cancer Research Hospital
Teams taking part in this year’s 2025 Cambridge Dragon Boat Festival have beat last year’s fundraising total to raise an impressive £40,000 for the new Cambridge Cancer Hospital.

Pioneering breast cancer programme opens in more hospitals across the East
Patients who join the Personalised Breast Cancer Programme, which was pioneered at Addenbrooke's Hospital in Cambridge, have their DNA read like a barcode, with the whole genome of their tumour sequenced and the results returned to inform treatment.

Social media star GK Barry visits Teenage Cancer Trust unit at CUH
Social media influencer and TV star Grace Keeling, known as GK Barry, visited Cambridge University Hospital’s NHS Foundation Trust (CUH) to spread some cheer to teenagers from the East of England region being treated for cancer.

Faster, more accessible clinical trial initiative being piloted in pancreatic cancer
The PemOla precision medicine trial in pancreatic cancer is exploring a new way of running clinical trials that can help more patients to get treatment closer to home.

Low-risk patients could avoid more aggressive breast cancer treatments, study shows
Targeted breast radiotherapy is as safe and effective as less focused approaches that have more side effects





